Research & Development
Applied liposomal development designed to translate formulations into controlled, scalable manufacturing processes.
GMPriority Pharma provides structured R&D services for partners requiring formulation refinement, feasibility assessment and controlled scale-up within defined manufacturing and regulatory frameworks.
______________________________________________________________
Our focus is not exploratory research, but applied development designed to support reliable commercial manufacture.
Development programmes are structured to identify formulation constraints early, define processing parameters and reduce scale-up risk.
Applied liposomal development

R&D at GMPriority Pharma is focused on practical execution, supporting the transition from formulation concept to manufacturable system.
R&D services may include:
- Feasibility assessment of lipid-based delivery systems
- Formulation optimisation (including lipid composition and encapsulation approach)
- Stability evaluation under defined storage and handling conditions
- Particle size distribution and structural characterisation (including PDI)
- Processing parameter definition (e.g. temperature, mixing, energy input)
- Translation from laboratory-scale to manufacturing conditions
All development activity is conducted with manufacturing integration in mind.
Development within controlled manufacturing frameworks
R&D is undertaken within the same operational environment that supports commercial production.
This supports:
→ Defined and transferable process documentation
→ Alignment with GMP-aligned manufacturing systems
→ Compatibility with cleanroom production environments
→ Scalable process design based on real equipment and constraints
Development parameters are established with future scale, batch consistency and supply continuity considered from the outset.

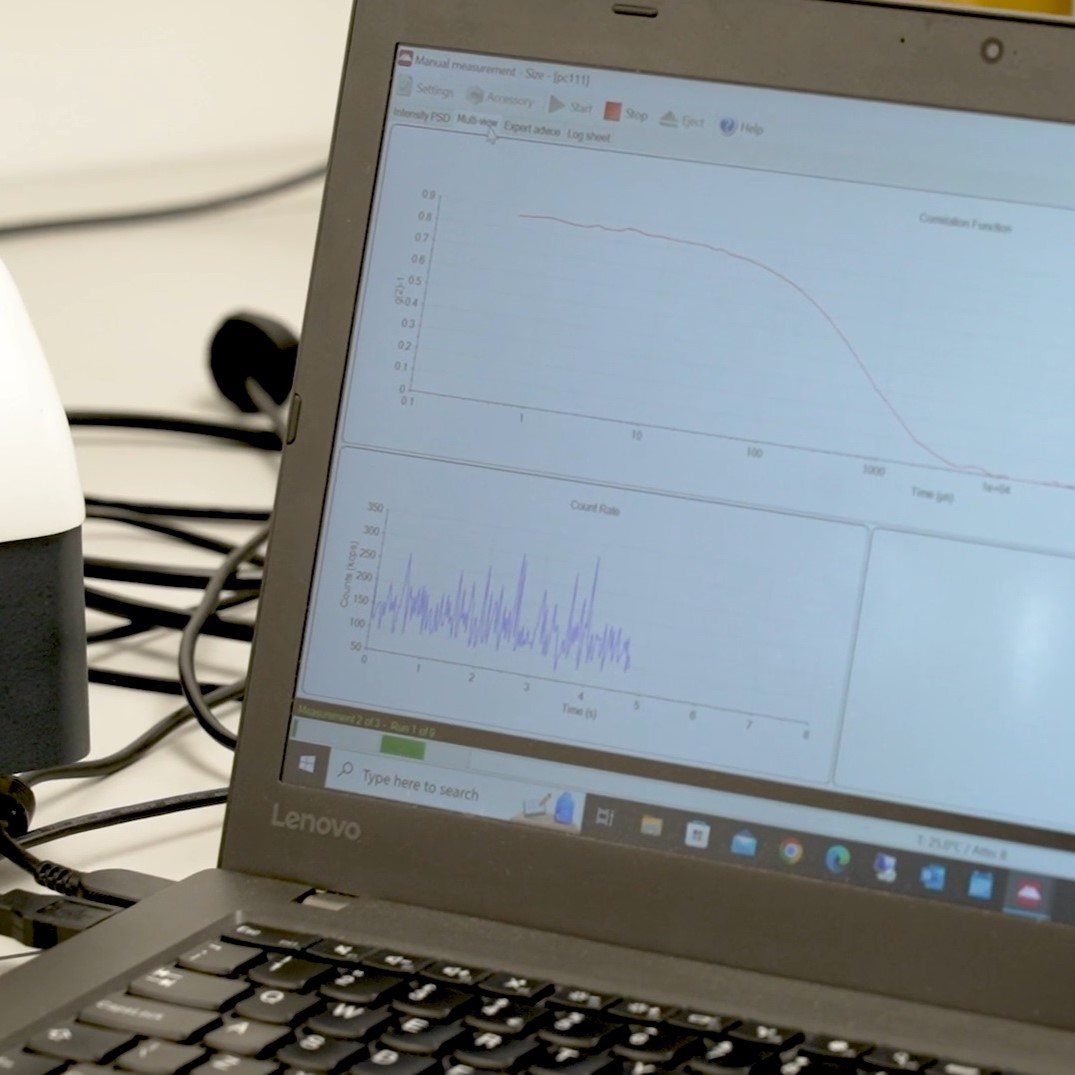

Characterisation & Evaluation
Development programmes are supported by in-house analytical capability, including:
- Particle size distribution analysis (e.g. DLS)
- Polydispersity index (PDI) assessment
- Dispersion stability monitoring under defined conditions
- Structural evaluation of liposomal systems
Characterisation data is used to inform formulation decisions, define acceptable parameter ranges and support reproducible processing.
Designed to reduce
Scale-up failure between lab and production
Batch variability linked to undefined parameters
Reformulation during transfer to manufacturing
Engaging with
GMPriority Pharma R&D
R&D services are appropriate for:
Ξ Brands developing new liposomal formulations requiring structured development
Ξ Ingredient partners seeking feasibility evaluation and optimisation
Ξ Programmes requiring scale-up support into commercial manufacture
Ξ Scientific collaborations aligned with defined commercial or production outcomes
Projects are assessed individually to ensure alignment with manufacturing capability, technical feasibility and long-term supply requirements.
Discuss scientific collaboration
Enquiries relating to formulation development, feasibility assessment or scale-up are considered on a case-by-case basis.
Please contact GMPriority Pharma with an outline of your proposed project.

